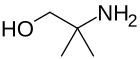
Alkanolamine
In organic chemistry, alkanolamines (amino alcohols) are organic compounds that contain both hydroxyl (−OH) and amino (−NH2, −NHR, and −NR2) functional groups on an alkane backbone. Alkanolamine's bifunctionality and physicochemical characteristics lead to its use in many applications, such as textiles, cosmetics, agricultural chemical intermediates, drugs, and metal working fluids.[1][2] Some aminoalcohols derivatives also have chemotherapeutic properties. [3]
- Alkanolamines
-
Methanolamine, from the reaction of ammonia with formaldehyde
-
2-amino-2-methyl-1-propanol is a precursor to oxazolines
-
Sphingosine is a component of some cell membrane.
Alkanolamines usually have high-solubility in water due to the hydrogen bonding ability of both the hydroxyl group and the amino group.[4] Alkanoamines have also shown a broad toxicity for a variety of organisms, including parasites, insect larvae and eggs, and microbes. Studies have also shown that the antimicrobial effect of alkanolamines increases in higher pH's. [5] Most alkanolamines are colorless.[6]
1-Aminoalcohols
[edit]1-Aminoalcohols are better known as hemiaminals. Methanolamine is the simplest member. 1-Aminoalcohols tend to be labile, readily converting to more highly condensed derivatives or hydrolyzing to the amine and carbonyl.
2-Aminoalcohols
[edit]2-Aminoalcohols, or 1,2-aminoalcohols are an important class of organic compounds. 2-aminoalcohols have been used in target molecule synthesis, chiral auxiliaries, or ligands.[3] This is because 2-aminoalcohols are able to create another stereogenic center.[3] 2-aminoalcohols are often generated by the reaction of amines with epoxides:
- C2H4O + R−NH2 → RNHC2H4OH
2-Aminoalcohols can also be synthesized from cyclic systems containing an oxygen and a nitrogen.[3] One popular method to synthesizing 2-aminoalcohols is the Sharpless asymmetric amino hydroxylation.[7]
Simple alkanolamines are used as solvents, synthetic intermediates, and high-boiling bases.[6]
Hydrogenation or hydride reduction of amino acids gives the corresponding 2-aminoalcohols. Examples include prolinol (from proline), valinol (from valine), tyrosinol (from tyrosine).
Key members: ethanolamine, dimethylethanolamine, N-methylethanolamine, Aminomethyl propanol. Two popular drugs, often called alkanolamine beta blockers, are members of this structural class: propranolol, pindolol.[8][9][10] 2-Aminoalcohols can also be found in the direct action subgroup of adrenergic drugs such as epinephrine, isoproterenol, phenylephrine and isoetarine.[11] Isoetarine is yet another medicinally useful derivative of ethanolamine.[citation needed]
1,3- to 1,7-amino alcohols
[edit]Here are two examples of longer aminoalcohols.
- Heptaminol, a cardiac stimulant
- Propanolamines
1,3-aminoalcohols are present in several bioactive molecules, such as Sedinone, Dumetorine, and Hygroline.[12] 1,3-aminoalcohols have be synthesize through a couple methods. Similar to 2-aminoalcohols, 1,3 aminoalcohols can be formed through ring openings, such as an azo-ring opening and addition.[12] 1,3-aminoalcohols can also be synthesized through an azo-aldol condensation or an intermolecular C-H activation.[12]
1,4 and 1,5-aminoalcohols have been synthesized through the reduction of cyclic amides.[13] Catalyzed alkylation of primary amines with 1,4-butanediol is another synthetic route for 1,4-aminoalcohols.[13] Larger amino alcohol (1,5 - and up) synthesis is comparatively underdeveloped. Recently, electrochemical ring-openings have shown robustness and versatility in producing 1,3 to 1,7-aminoalcohols. [14]
Natural Products & Drugs
[edit]Alkanolamines are present in biomolecules and building blocks leading them to be present in most proteins and peptides. There are 88 approved drugs and more than 3600 natural products that contain aminoalcohols.[7] Two amino acids are alkanolamines, formally speaking: serine and hydroxyproline.
- Veratridine and veratrine
- Tropane alkaloids such as atropine
- hormones and neurotransmitters epinephrine (adrenaline) and norepinephrine (noradrenaline)
The electrochemical ring-opening synthesis has show compatibility with natural aliphatic amino acids, including serine.[14] Amino ethanols have been proven to be vital precursors for chiral morpholines and piperazines.[7]
References
[edit]- ^ Davis, John W.; Carpenter, Constance L. (1997), Ware, George W. (ed.), "Environmental Assessment of the Alkanolamines", Reviews of Environmental Contamination and Toxicology, 149, New York, NY: Springer New York: 87–137, doi:10.1007/978-1-4612-2272-9_2, ISBN 978-1-4612-7482-7, PMID 8956559, retrieved 2025-03-19
- ^ Laskar, Ranjini; Dutta, Subhabrata; Spies, Jan C.; Mukherjee, Poulami; Rentería-Gómez, Ángel; Thielemann, Rebecca E.; Daniliuc, Constantin G.; Gutierrez, Osvaldo; Glorius, Frank (2024-04-17). "γ-Amino Alcohols via Energy Transfer Enabled Brook Rearrangement". Journal of the American Chemical Society. 146 (15): 10899–10907. Bibcode:2024JAChS.14610899L. doi:10.1021/jacs.4c01667. ISSN 0002-7863. PMC 11027157. PMID 38569596.
- ^ a b c d Ager, David J.; Prakash, Indra; Schaad, David R. (1996-01-01). "1,2-Amino Alcohols and Their Heterocyclic Derivatives as Chiral Auxiliaries in Asymmetric Synthesis". Chemical Reviews. 96 (2): 835–876. doi:10.1021/cr9500038. ISSN 0009-2665.
- ^ "Amino Alcohols - Alfa Chemistry". www.alfa-chemistry.com. Retrieved 2025-03-28.
- ^ Sandin, M; Allenmark, S; Edebo, L (March 1990). "Selective toxicity of alkanolamines". Antimicrobial Agents and Chemotherapy. 34 (3): 491–493. doi:10.1128/AAC.34.3.491. ISSN 0066-4804. PMC 171625. PMID 2334165.
- ^ a b Martin Ernst; Johann-Peter Melder; Franz Ingo Berger; Christian Koch (2022). "Ethanolamines and Propanolamines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a10_001.pub2. ISBN 978-3-527-30673-2.
- ^ a b c Sun, Jiawei; Wang, Shuanghu; Harper, Kaid C.; Kawamata, Yu; Baran, Phil S. (January 2025). "Stereoselective amino alcohol synthesis via chemoselective electrocatalytic radical cross-couplings". Nature Chemistry. 17 (1): 44–53. doi:10.1038/s41557-024-01695-7. ISSN 1755-4349.
- ^ "Propranolol Monograph for Professionals". Drugs.com. Retrieved 2025-03-28.
- ^ "Pindolol Uses, Side Effects & Warnings". Drugs.com. Retrieved 2025-03-28.
- ^ Wong, Gavin W. K.; Boyda, Heidi N.; Wright, James M. (2014-11-27). "Blood pressure lowering efficacy of partial agonist beta blocker monotherapy for primary hypertension". The Cochrane Database of Systematic Reviews. 2014 (11): CD007450. doi:10.1002/14651858.CD007450.pub2. ISSN 1469-493X. PMC 6486122. PMID 25427719.
- ^ Vardanyan, R. S.; Hruby, V. J. (2006-01-01), Vardanyan, R. S.; Hruby, V. J. (eds.), "11 - Adrenergic (Sympathomimetic) Drugs", Synthesis of Essential Drugs, Amsterdam: Elsevier, pp. 143–159, ISBN 978-0-444-52166-8, retrieved 2025-03-28
- ^ a b c Wang, Wei; Hu, Yi; Lin, Ruiqi; Wu, Heng; Tong, Qi; Wang, Liansheng; Xiao, Zufeng; Zhu, Lei (2020). "Progress on the Synthesis of 1,3-Amino Alcohol". Chinese Journal of Organic Chemistry. 40 (5): 1129. doi:10.6023/cjoc201911011. ISSN 0253-2786.
- ^ a b Xiao, Zhen; Li, Juanjuan; Yue, Qiang; Zhang, Qian; Li, Dong (2018). "An efficient and atom-economical route to N -aryl amino alcohols from primary amines". RSC Advances. 8 (60): 34304–34308. doi:10.1039/C8RA07355D. ISSN 2046-2069. PMC 9086943. PMID 35548644.
- ^ a b Fang, Xinyue; Hu, Xinwei; Li, Quan-Xin; Ni, Shao-Fei; Ruan, Zhixiong (2025). "Paired Electro-Synthesis of Remote Amino Alcohols with/in H2O". Angewandte Chemie International Edition. 64 (6): e202418277. doi:10.1002/anie.202418277. ISSN 1521-3773.
External links
[edit]- Amino+Alcohols at the U.S. National Library of Medicine Medical Subject Headings (MeSH)